|
Infective Endocarditis:
Epidemiology and Pathogenesis: IE predominantly affects individuals with predisposing cardiac abnormalities, such as congenital heart diseases, rheumatic heart diseases, prosthetic heart valves, or intracardiac devices. Intravenous drug abuse, indwelling catheters, poor dental hygiene, and immunocompromised states like HIV also increase the risk. The pathogenesis involves the formation of a platelet-fibrin vegetation on the endocardial surface, which serves as a nidus for bacterial colonization and proliferation, leading to tissue destruction and potential embolization. Causative Organisms: Staphylococcus aureus is the most common causative pathogen, accounting for approximately 30% of cases. Viridans group streptococci, enterococci, and coagulase-negative staphylococci are other frequently implicated microorganisms. The HACEK group (Haemophilus, Actinobacillus, Cardiobacterium, Eikenella, and Kingella) and fungi (Candida species) are less common etiologies. Clinical Manifestations: The clinical presentation of IE can be acute or subacute, depending on the virulence of the pathogen and the host's immune status. Common manifestations include fever, chills, malaise, anorexia, and weight loss. Cardiac manifestations may include new or changing murmurs, heart failure, or embolic events. Non-specific symptoms like arthralgia, myalgia, and back pain may also occur. The Duke Criteria: The Duke Criteria, established in 1994 and later modified in 2000, are widely used for the diagnosis of IE. These criteria incorporate clinical, microbiological, and echocardiographic findings. The diagnosis is confirmed by meeting two major criteria, one major and three minor criteria, or five minor criteria. Major Criteria include: 1. Positive blood cultures for IE pathogens. 2. Evidence of endocardial involvement on echocardiography, such as vegetations, abscess, or new valvular regurgitation. Minor Criteria include: 1. Predisposing cardiac conditions or intravenous drug abuse. 2. Fever. 3. Vascular phenomena, such as embolic events, Janeway lesions, etc. 4. Immunologic phenomena, such as glomerulonephritis, Osler nodes, Roth spots, etc. 5. Microbiological evidence not meeting major criteria. The Duke Criteria incorporate clinical, microbiological, and echocardiographic findings. The criteria demonstrate a sensitivity exceeding 90% for the diagnosis of IE. Management Principles: Early empiric antibiotic therapy is crucial, with vancomycin being the most appropriate choice for suspected IE due to its broad coverage of gram-positive organisms, including methicillin-resistant Staphylococcus aureus (MRSA). Subsequent antibiotic regimens should be tailored based on blood culture results and susceptibility patterns. Surgical intervention may be necessary in cases of refractory heart failure, uncontrolled infection, or high risk of embolization. Indications for surgery include valve dysfunction, perivalvular extension, or persistent bacteremia despite appropriate antimicrobial therapy. Prevention and Prophylaxis: Strict adherence to sterile techniques during invasive procedures and prompt management of bacteremia are crucial preventive measures. Antibiotic prophylaxis is recommended for individuals with specific high-risk cardiac conditions undergoing dental procedures or procedures involving manipulation of infected tissues.
0 Comments
Pitfalls of pulse oximetry
In this lecture, we will discuss the pitfalls of using pulse oximeter in assessing patients with dyspnea. Pulse oximetry is a non-invasive method of measuring the oxygen saturation of hemoglobin in arterial blood. It has become a ubiquitous tool in clinical practice, providing a continuous estimate of a patient's oxygenation status. However, there are several important limitations and potential pitfalls associated with the use of pulse oximetry, particularly in the assessment of patients presenting with dyspnea or respiratory distress. The fundamental principle behind pulse oximetry involves the different absorption spectra of oxyhemoglobin and deoxyhemoglobin for red and infrared light. The ratio of these absorption values is used to calculate the oxygen saturation (SpO2). This calculation assumes that only two species of hemoglobin are present: oxyhemoglobin and deoxyhemoglobin. However, in certain clinical situations, other hemoglobin species may be present, leading to inaccurate SpO2 readings. One such situation is carbon monoxide poisoning. Carboxyhemoglobin, formed by the binding of carbon monoxide to hemoglobin, has a similar absorption spectrum to oxyhemoglobin. Since oximeters misinterpret carboxyhemoglobin as 100% oxyhemoglobin, SpO2 will trend toward 100% in patients with significant carbon monoxide poisoning, potentially masking the severity of their condition. This can lead to a false sense of reassurance and delayed treatment. Another limitation of pulse oximetry is its inability to detect hypoxemia in the setting of abnormal hemoglobin species, such as methemoglobin and sulfhemoglobin. Methemoglobinemia can occur due to certain drugs, toxins, or inherited disorders, while sulfhemoglobinemia is typically associated with sulfur compound exposure. Sulfhemoglobinemia can mimic methemoglobinemia clinically and by SpO2 measurements. Since oximeters misinterpret methemoglobinemia as 85% oxyhemoglobin, SpO2 will trend toward 85% in patients with significant methemoglobinemia. Thus, SpO2 can either overestimate or underestimate the oxygen saturation of patients with methemoglobinemia or sulfhemoglobinemia. Furthermore, pulse oximetry does not consider hemoglobin level, and has limitations in accurately measuring oxygen saturation at extremes of hemoglobin concentration. For non-hypoxic cases of severe anemia with hemoglobin levels below 5 g/dL, pulse oximeters may overestimate the oxygen saturation. Conversely, in patients with polycythemia or high hemoglobin levels, pulse oximeters may underestimate the oxygen saturation. It is also important to note that pulse oximetry measures the functional saturation of hemoglobin, which may not accurately reflect the overall oxygen content of the blood, particularly in cases of dyshemoglobinemias or shifts in the oxygen-hemoglobin dissociation curve. Pulse oximetry does not consider A-a gradient, supplemental O2 can mask severe pulmonary process. In such situations, arterial blood gas analysis provides a more comprehensive assessment of oxygenation status and acid-base balance. Fluctuations in PaO2 can lead to rapid, unpredictable changes in SpO2 that may be missed by intermittent monitoring, thus, continuous SpO2 monitoring is suggested for dyspneic patients. In addition to these limitations, pulse oximetry can be affected by factors such as poor perfusion, motion artifacts, and the presence of certain dyes or pigments. Conditions such as hypothermia, hypotension, or peripheral vascular disease can lead to reduced peripheral perfusion, resulting in unreliable SpO2 readings. Similarly, excessive patient movement can introduce motion artifacts, compromising the accuracy of the measurements. Another pitfall is that pulse oximetry measures peripheral oxygen saturation, which can lag behind changes in central arterial oxygenation. This delay may mask acute deterioration in some patients. When assessing patients with dyspnea or respiratory distress, it is crucial to interpret pulse oximetry readings in the context of the patient's clinical presentation, physical examination findings, and other diagnostic modalities. Relying solely on pulse oximetry can potentially lead to missed diagnoses or inappropriate management decisions. Arterial Blood Gas (ABG) provides a more comprehensive assessment of oxygenation and acid-base balance. ABG allows the calculation of A-a gradient which helps clinicians narrow down the differential diagnosis for hypoxemia. One should consider ABG analysis when appropriate. In summary, while pulse oximetry is a valuable tool in clinical practice, healthcare professionals must be aware of its limitations, particularly in the assessment of patients with dyspnea or respiratory distress. Potential pitfalls include interference from abnormal hemoglobin species, inaccuracies at extremes of hemoglobin concentration, the inability to detect hypoxemia in certain conditions, and the effects of poor perfusion or motion artifacts. Integrating pulse oximetry findings with a comprehensive clinical evaluation, including arterial blood gas analysis when appropriate, is essential for accurate diagnosis and appropriate management of respiratory disorders. Take Home Message
Back pain Emergencies
Aortic syndrome or retroperitoneal bleeding
Spinal epidural abscess (SEA)
Cauda equina syndrome
Straight Leg Raise Test (SLRT)
The slump test
Spinal Metastases
Spinal Fractures
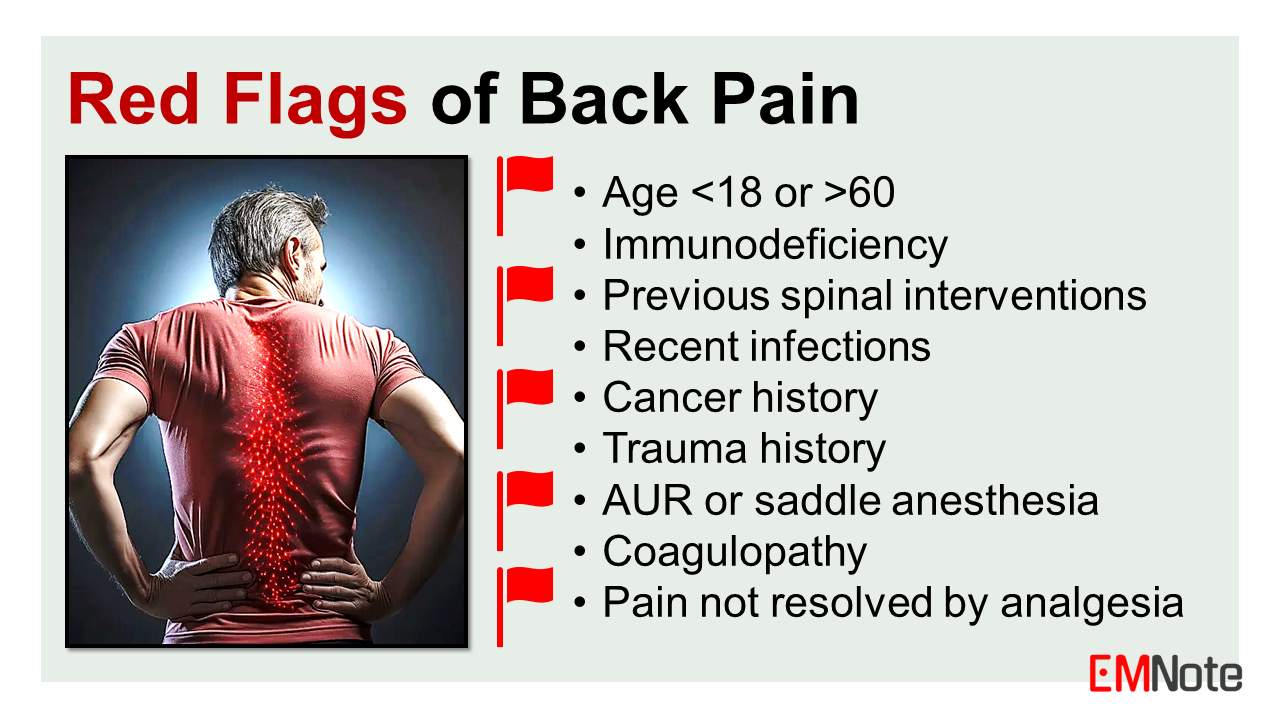
Red Flags for Back Pain
Important Physical Findings of patients with back pain
Diagnostic Tests for Back Pain
Management of Back Pain Emergencies
Take Home Message
EM Literature #1
- MgS04 for rapid A-fib - NaHC03 for DKA - Norepinephrine for post-ROSC - Antihistamines for vertigo - Contrast nephropathy - NTG for RV MI - Less fluid for pancreatitis - High-dose NTG for SCAPE |

Author
|
Proudly powered by Weebly